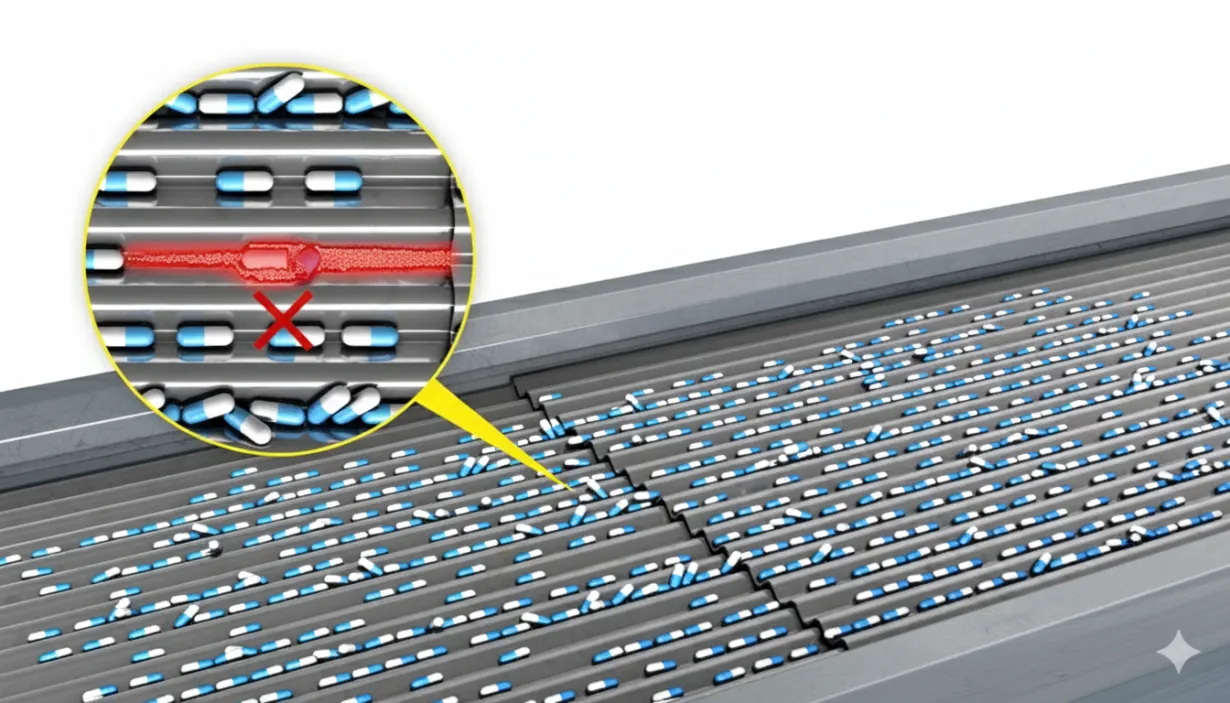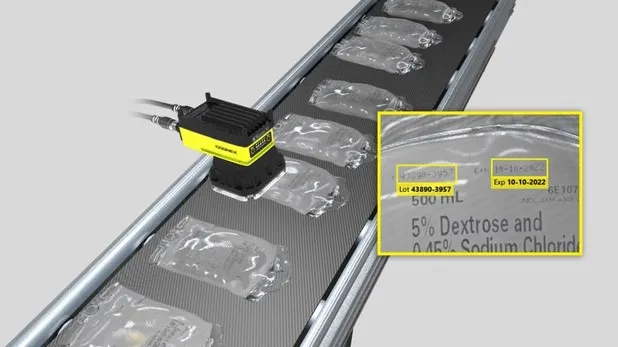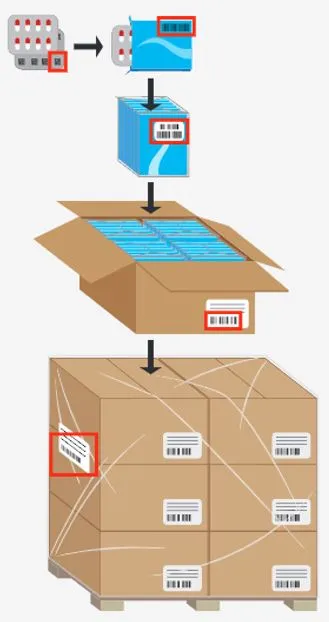
To comply with current pharmaceutical traceability and anti-counterfeiting regulations, each carton must be marked with a unique serial number (serialization), and in some cases, each aggregation must be labeled with a barcode indicating the products contained (aggregation).
To provide a modular and scalable solution that meets current regulations and is easily adaptable to future requirements, the TRZ product has been developed. Through its various modules, TRZ provides all the necessary elements for line-level compliance and the ability to scale to plant and corporate levels.
TRZ enables complete traceability process control, including:
- Commissioning and decommissioning of items.
- Automatic and manual aggregations.
- Modifications during a running batch.
- Management of peak cartons and leftover batch items.
- Real-time status consultation of cartons or pallets.
- Batch incident resolution.
Standard Equipment Features:
- Software designed to ensure compliance with total pharmaceutical traceability regulations (EU, USA, Korea, China, Turkey, Brazil, etc.).
- Verification of different parameters based on the traceability type (fixed code, incremental code, repetition control, out-of-range products, etc.).
- Centralized input of batch data (lot number, expiration date, etc.) for distribution to various devices (printer, inspection system, etc.).
- Ability to add or remove products via manual barcode reader.
- Production reconciliation at the end of the batch.
- Real-time production data storage in a relational database.
- Export capability to external databases.
- Generation of batch reports exportable in multiple formats (PDF, XML, etc.).
- Access to batch data through the operator panel. Data security ensured via backup and restore functions.